Adsorption of Methyl Orange from Aqueous Solution using Ni/Al LDH Modified with Camellia sinensis Leaf Extracts
DOI:
https://doi.org/10.26554/ijmr.20264393Keywords:
Ni/Al LDH, Camellia sinensis, Composite, Adsorption, Methyl OrangeAbstract
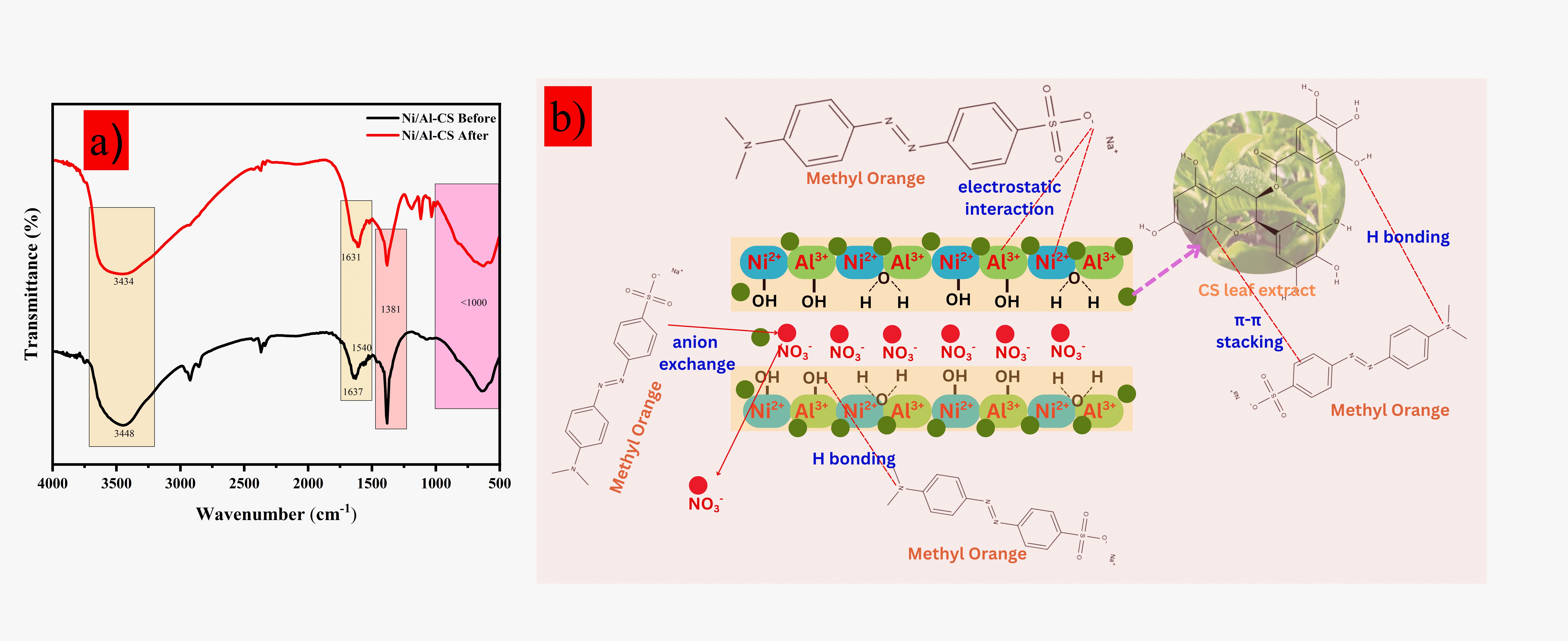
Water pollution caused by dye pollutants such as methyl orange (MO) can have a negative impact on humans, living organisms, and ecosystems. Adsorption is one of the promising methods in overcoming the presence of MO pollutants. This research focuses on the synthesis of layered double hydroxide (LDH) Ni/Al composites prepared by coprecipitation method with the addition of green tea/Camellia sinensis (CS) leaf extract as supporting material. The synthesized materials obtained were then characterized using X-ray diffraction (XRD) patterns and Fourier transform infrared spectra (FTIR). The Ni/Al and Ni/Al-CS materials were then evaluated as adsorbents to adsorb MO from aquatic solutions. The maximum capacity of MO adsorption obtained was 18.519 mg.g−1 on Ni/Al LDH and 49.261 mg.g−1 on Ni/Al-CS, respectively. The Langmuir isotherm model showed the best fit to the adsorption data on both materials, while the kinetics of the adsorption process followed a pseudo second-order (PSO) model. Thermodynamic analysis (ΔG°, ΔS°, and ΔH°) showed that the MO adsorption process on both materials was spontaneous and endothermic. The regeneration process carried out four consecutive regeneration cycles showed that Ni/Al-CS material has excellent adsorbent recycling ability, which only decreased by 10.26%. In contrast to Ni/Al LDH which experienced a significant decrease of up to 31.70% in the 4th cycle. These findings suggest that Ni/Al CS material is a promising adsorbent for MO removal applications from aquatic solutions.
References
Abbou, B., I. Lebkiri, H. Ouaddari, A. El Amri, F. E. Achibat, L. Kadiri, A. Ouass, A. Lebkiri, and E. H. Rifi (2023). Improved Removal of Methyl Orange Dye by Adsorption Using Modified Clay: Combined Experimental Study Using Surface Response Methodology. Inorganic Chemistry Communications, 155; 111127
Abd Al-khuder, Z. H. and F. F. Karam (2025). Synthesis and Characterization of a Quaternary Composite Based on RGO/MWCNTs/Choline Chloride + Malonic Acid for Methyl Orange Dye Adsorption. Results in Chemistry, 15; 102133
Ahmad, N., S. P. J. Negara, et al. (2024a). Insight of Anionic Dyes Adsorption from Their Aqueous Solutions onto MgAl LDH/Lignin: Characterization and Isotherm Studies. Indonesian Journal of Material Research, 2(2); 40–46
Ahmad, N., A. Wijaya, A. Lesbani, et al. (2024b). Magnetite Humic Acid-Decorated MgAl Layered Double Hydroxide and Its Application in Procion Red Adsorption. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 684; 133042
Ahmad, N., A. Wijaya, et al. (2025). Comparative Assessment of Procion Red Removal Using Magnetite-Based Composites with Humic Acid, Activated Charcoal, and Lignin. Indonesian Journal of Material Research, 3(3); 85–92
Alsaab, H. O., S. Shirazian, N. Pirestani, and R. Soltani (2025). Sustainable Synthesis and Dual Adsorption of Methyl Orange and Cadmium Ions Using Biogenic Silica-Based Fibrous Silica Functionalized with Crown Ether Ionic Liquid. Journal of Colloid and Interface Science, 679; 555–568
Alyasi, H., H. Mackey, and G. McKay (2023). Adsorption of Methyl Orange from Water Using Chitosan Bead-Like Materials. Molecules, 28(18); 6561
Amri, A., A. Lesbani, and R. Mohadi (2023). Malachite Green Dye Adsorption from Aqueous Solution Using a Ni/Al Layered Double Hydroxide-Graphene Oxide Composite Material. Science and Technology Indonesia, 8(2); 280–287
Amri, A., S. Wibiyan, A. Wijaya, N. Ahmad, R. Mohadi, and A. Lesbani (2024). Efficient Adsorption of Methylene Blue Dye Using Ni/Al Layered Double Hydroxide-Graphene Oxide Composite. Bulletin of Chemical Reaction Engineering & Catalysis, 19(2); 181–189
Borah, A. R., M. Gogoi, R. Goswami, P. K. Nath, and S. Hazarika (2025). Chiral Ni-Al LH Nanoparticle Embedded Electrospun Nanofibrous Membrane with High and Stable Permeance for Enantioseparation of Ketoprofen. Separation and Purification Technology, 361; 131443
Boucif, F., F. Bessaha, F. Bendahma, G. Bessaha, N. Mahrez, M. Sillanpää, and A. Khelifa (2025). Facile Synthesis and Modification of Layered Double Hydroxides (LDH) for Adsorption of Orange I and Acid Red 114 Dyes from Aqueous Solution: Performance and Mechanism Study. Inorganic Chemistry Communications, 178; 114673
Dehmani, Y., D. S. Franco, J. Georgin, R. Mghaiouini, B. B. Mohammed, R. Kacimi, T. Lamhasni, E. C. Lima, N. El Messaoudi, and A. Sadik (2025). Towards a Deeper Understanding of the Adsorption of Methyl Orange on a Commercial Activated Carbon: Study of Impact Factors, Isotherm and Mechanism. Environmental Surfaces and Interfaces, 3; 103–111
Dong, Y., J. Liang, J. Song, C. Liu, Z. Ding, W. Wang, W. Zhang, et al. (2024). Preparation of Biochar/Iron Mineral Composites and Their Adsorption of Methyl Orange. RSC Advances, 14(46); 33977–33986
Duan, J., S. Liu, X. Xu, Y. Zhang, Z. Dong, Z. Nie, R. Liu, X. Ren, and B. Wang (2025). Alginate Aerogel with High Adsorption Performance for Copper Ions. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 719; 137054
Duddi, R., S. Dhiman, A. K. Singh, N. Kamboj, S. Kumar, et al. (2025). Hydrothermally Synthesised Porous NiAl-LDH As an Efficient Pseudocapacitive Material in Asymmetric Supercapacitors. Hybrid Advances, 8; 100372
El Foulani, A.-A., S. Tarhouchi, I. Hammoudan, R. Saddik, A. Y. A. Alzahrani, and S. Tighadouini (2025). A Novel Hydrazide-Modified Silica Gel As a Promising Material for Adsorption of Methyl Orange from Aqueous Solutions: Experiments and Theoretical Insights. Results in Surfaces and Interfaces, 18; 100455
Etim, E. E., S. Yakubu, A. Terhembe, and L. J. Moses (2024). Investigations on the Biosorption of Nickel Using Tea Leaves and Tea Fibre (Camellia sinensis) As Adsorbents: Thermodynamics, Isotherms and Kinetics. Discover Chemistry, 1(1); 3
Fu, S., Z. Xie, R. Wang, H. Zou, S. Lian, and R. Guo (2023). Combined Disposal of Methyl Orange and Corn Straw Via Stepwise Adsorption-Biomethanation-Composting. Journal of Environmental Management, 344; 118358
Hammood, Z. A. and A. A. Mohammed (2024). Adsorption of Tetracycline from an Aqueous Solution on a CaMgAl-Layer Double Hydroxide/Red Mud Composite: Kinetic, Isotherm, and Thermodynamic Studies. Environmental Nanotechnology, Monitoring & Management, 22; 101018
Han, J. H., D. H. Keum, V. Kothuri, Y.-J. Kim, H. C. Kwon, D. H. Kim, H. S. Jung, and S. G. Han (2024). Enhancing Emulsion, Texture, Rheological and Sensory Properties of Plant-Based Meat Analogs with Green Tea Extracts. Food Chemistry: X, 24; 101807
Hariprasath, K., M. Priyadharshini, P. Balaji, R. Thangappan, T. Pazhanivel, et al. (2024). Electrode Engineering Strategies for Boosting the Performance of NiCoAl-LDH in Supercapacitor Application. Chemical Physics Letters, 856; 141584
Hashem, M. A., S. F. Rahman, S. R. Sium, M. Maoya, M. M. Miem, A. A. Mimi, and M. E. H. Zahin (2025). Thermally Activated Adsorbent Derived from Kitchen Biowaste for Treatment of Tannery Wastewater. Cleaner Chemical Engineering, 11; 100156
Hezari, S., A. Olad, and A. Dilmaghani (2022). Modified Gelatin/Iron-Based Metal-Organic Framework Nanocomposite Hydrogel As Wound Dressing: Synthesis, Antibacterial Activity, and Camellia Sinensis Release. International Journal of Biological Macromolecules, 218; 488–505
Hosseinpour, E. and A. Rahbar-Kelishami (2025). Cationic Surfactant Modified NaY Zeolite: Preparation, Investigating the Effect of Surfactant Concentration, and Application for Methyl Orange Adsorption from Aqueous Solution. Journal of Environmental Chemical Engineering, 13(3); 116406
Jin, C., J. Yang, R. Zuo, W. Li, J. Wang, and J. Xu (2025). Adsorption Behavior and Mechanism of MnMgAl-Layered Double Hydroxide for the Removal of V(V): Experiments and DFT Calculation. Process Safety and Environmental Protection, 198; 107187
Leng, Y., J. Li, J. Liu, F. Chang, Z. Li, Y. Huang, W. Xiong, B. Wu, B. Han, H. Chen, et al. (2025). Adsorption of Methyl Parathion on Four Various Microplastics in Aqueous Solution: Kinetics, Isotherms and Molecular Dynamics Simulations. Gondwana Research, 144; 33–48
Lesbani, A., N. Ahmad, R. Mohadi, I. Royani, S. Wibiyan, Y. Hanifah, et al. (2024). Selective Adsorption of Cationic Dyes by Layered Double Hydroxide with Assist Algae (Spirulina platensis) to Enrich Functional Groups. JCIS Open, 15; 100118
Li, Y.-J., T.-T. Qi, Y.-N. Dong, W.-H. Hou, G.-W. Chu, L.-L. Zhang, and B.-C. Sun (2022). Synthesized Ni/MMO Catalysts via Ultrathin Ni-Al-LDH in a Rotating Packed Bed for Hydrogenation of Maleic Anhydride. Fuel, 326; 125035
Ma, Q., X. Han, J. Cui, Y. Zhang, and W. He (2022). Ni Embedded Carbon Nanofibers/Ni-Al LDHs with Multicomponent Synergy for Hybrid Supercapacitor Electrodes. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 649; 129270
Martins, N. J. and O. W. Perez-Lopez (2025). Tuning the Composition of Ni-Al-LDH Catalysts for Low-Temperature CO2 Methanation. Fuel, 381; 133594
Milovanovic, S., I. Lukic, N. Krgovic, V. Tadic, Z. Radovanovic, K. Tyśkiewicz, and M. Konkol (2024). Selection of Processing Parameters for the Integrated Supercritical CO2 Extraction from Green Tea Leaves and Extract Impregnation onto Starchchitosan Based Films. The Journal of Supercritical Fluids, 206; 106163
Mishra, A., D. Singh, R. S. Singh, V. Mishra, M. Kumar, and B. S. Giri (2024). Performance Study of the Bioreactor for the Biodegradation of Methyl Orange Dye by Luffa Immobilized Stenotrophomonas Maltophilia and Kinetic Studies: A Sustainable Approach. Groundwater for Sustainable Development, 27; 101378
Mubarak, M. F., T. A. Yousef, S. A. Salim, M. Khairy, E. A. Kamoun, and T. Mahmoud (2025). Meta-Kaolinite Metal Oxide Quaternary Composite for Layered Double Hydroxide Applied to a New Frontier in Adsorption Technology: Synthesis, Adsorption Performance and Kinetics Study. Inorganic Chemistry Communications, 178; 114647
Pan, X., N. Zhao, H. Shi, H.Wang, F. Ruan, H.Wang, and Q. Feng (2025). Biomass Activated Carbon Derived from Golden Needle Mushroom Root for the Methylene Blue and Methyl Orange Adsorption from Wastewater. Industrial Crops and Products, 223; 120051
Popat, P. R., A. Y. Alyami, G. K. Inwati, B. A. Makwana, M. A. Alreshidi, J. S. Algethami, M. Abbas, and K. K. Yadav (2024). Enriched Adsorption of Methyl Orange by Zinc Doped Lithium Manganese Oxides Nanosorbent. Inorganic Chemistry Communications, 161; 112016
Purbasari, A., D. Ariyanti, and E. Fitriani (2023). Adsorption of Methyl Orange Dye by Modified Fly Ash-based Geopolymer–Characterization, Performance, Kinetics and Isotherm Studies. Journal of Ecological Engineering, 24(3); 90–98
Qiu, Z., Y. Zhang, X. Lv, and J. Dang (2025). High-Capacity Biomass-based Composite Aerogel for Efficient Multi-Ion Adsorption of Vanadium, Molybdenum, and Nickel. Separation and Purification Technology, 368; 132990
Ramutshatsha-Makhwedzha, D., A. Mavhungu, M. L. Moropeng, and R. Mbaya (2022). Activated Carbon Derived from Waste Orange and Lemon Peels for the Adsorption of Methyl Orange and Methylene Blue Dyes from Wastewater. Heliyon, 8(8); e09930
Roy, N., S. A. Alex, N. Chandrasekaran, K. Kannabiran, and A. Mukherjee (2022). Studies on the Removal of Acid Violet 7 Dye from Aqueous Solutions by Green ZnO@Fe3O4 Chitosan–Alginate Nanocomposite Synthesized Using Camellia sinensis Extract. Journal of Environmental Management, 303; 114128
Sajai, N., B. Soubai, S. El Khaider, A.-i. Chham, R. Fakhreddine, I. Mechnou, and S. Krimi (2025). Valorization of Calcium Phosphate Glasses: A Sustainable and Eco-Friendly Approach to Methylene Blue Dye Adsorption from Wastewater. Results in Surfaces and Interfaces, 19; 100525
Tcheka, C., M. M. Conradie, V. A. Assinale, and J. Conradie (2024). Mesoporous Biochar Derived from Egyptian Doum Palm (Hyphaene thebaica) Shells As Low-cost and Biodegradable Adsorbent for the Removal of Methyl Orange Dye: Characterization, Kinetic and Adsorption Mechanism. Chemical Physics Impact, 8; 100446
Waheed, T., P. Min, S. Shujaat, S. Haq, S. ud Din, M. K. Hossain, F. U. Rehman, A. Syed, A. H. Bahkali, L. S. Wong, et al. (2024). Enhanced Photocatalytic Performance of Cr/Ni/Mg/Al Layered Double Hydroxides Against Methyl Orange in Aqueous Solution. Desalination and Water Treatment, 319; 100495
Wibiyan, S., I. Royani, N. Ahmad, and A. Lesbani (2024). Assessing the Efficiency, Selectivity, and Reusability of ZnAl-layered Double Hydroxide and Eucheuma Cottonii Composite in Removing Anionic Dyes from Wastewater. Inorganic Chemistry Communications, 170; 113347
Wijaya, A., N. Ahmad, L. Hanum, E. Melwita, and A. Lesbani (2025). Spirogyra sp. Macro-Algae-Supported NiCr-LDH Adsorbent for Enhanced Remazol Red Dye Removal. Results in Surfaces and Interfaces, 18; 100427
Xie, C.-r., Y.-x. Song, G. Yang, C.-g. Sun, X. Luo, and T. Wu (2024). Porous Carbon Derived from MOF-235 for the Adsorption of Methyl Orange with High Capacity. Materials Today Communications, 41; 110843
Yang, P., J. Bi, H. Zhang, and Z. Wu (2024). EDTA-Interlayer Modified Mg/Fe Layered Double Hydroxides for Enhanced Adsorption of Methyl Orange: Adsorption Performance and Mechanism Study. Process Safety and Environmental Protection, 186; 1387–1396
Zhang, Q., Z. Wu, and S. He (2025). Preparation of Superhydrophilic and Underwater Superoleophobic Composite Graphene Aerogels and Their Performance in Dye Adsorption and Oil/Water Separation. Carbon, 241; 120384
Zhou, A., L. Zhu, Y. Chen, J.Wang, and Y. Liu (2025). Fast Adsorption of Low-concentration Ammonia Nitrogen by Persulfate-Modified Carbon Materials: Structure Influence, Performance, and Mechanism. Environmental Research, 278; 121680